Article: Lessons Learned in the Evolution of Advanced Therapy Logistics
By World Courier
At the recent ARM Meeting on the Mesa, I had the privilege of chairing a workshop that focused on “Evolving the Supply Chain for Advanced Therapies”.
The workshop involved Sven Kili, James Kovach, Thomas Fellner, Amy DuRoss and myself – all experienced cell and gene therapy professionals. The aim of the session was to use the combined speakers’ experience to identify the upcoming challenges posed by advanced therapies and provide the opportunity to discuss the lessons learned in what has been described as ‘the most complex logistics workflow and related requirements in the history of biologics’.
In an earlier session Bluebird CEO, Jeff Walsh perfectly summarized the industry by saying, “this “expletive” is hard”. Jeff’s rather blunt statement epitomizes in just a few words the complex challenges posed by advanced therapies within specialty logistics platforms, the most renowned of which are shown below in figure 1.
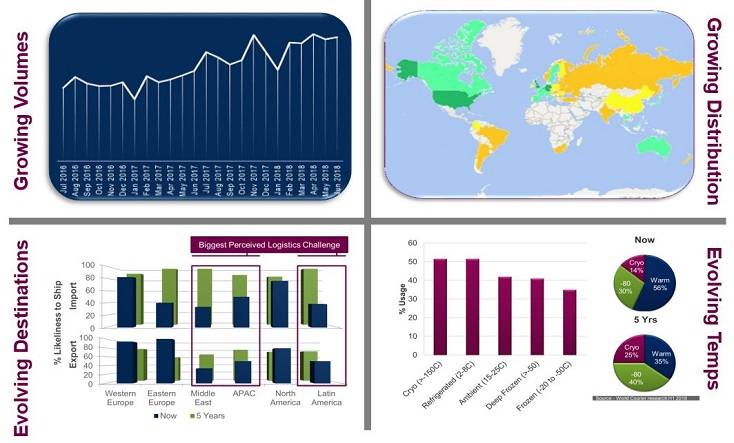
Figure 1
From these challenges 4 key lessons emerged: -
1) Plan early
Throughout the session the concept of thinking ahead was continually reinforced, as well as the emerging Logistics by Design concept which has evolved from Quality by Design principles. This provides a framework to identify high-risk areas that can then be used to develop a logistics platform.
2) Every question is relevant
Advanced therapy supply chains are complex with multiple moving parts across multiple organizations. Because of this we cannot assume that everybody knows everything, as there are not enough therapies in the healthcare system to have repeatable processes in place.
3) Consider standardization
This comment applies to almost every facet of the supply chain, from defining where therapeutic deliveries should be made in clinical sites (e.g. the pharmacy, the blood bank or the surgery) to agreeing to testing protocols for monitoring samples.
Standardization work is ongoing however companies need to consider what Intellectual Property needs to be protected and what would could be shared through groups like the Standards Coordinating Body to help the industry move forwards by creating consistency.
4) Use vendors as technical experts
Including the chair there were five vendors on this panel. They were invited to speak, not because they offered services at competitive prices, but because they were technical experts with valuable insight that should be harnessed by therapy developers to help them build, test and optimize their logistics platforms.
If you want to know more about the discussion please contact me via sellison@worldcourier.co.uk? or view the recording.




