Building precision into radiopharmaceutical logistics
By World Courier


“Radiopharmaceutical logistics is unlike anything else in healthcare supply chains,” says Andrea Zobel, Senior Director, Strategy, World Courier. “You’re balancing radioactive safety, pharmaceutical integrity, and extreme time pressure all at once. There’s no margin for error — every shipment represents a real patient waiting for diagnosis or treatment.”
What are radiopharmaceuticals? A brief introduction
Nuclear medicine, which uses radioactive materials for the research, diagnosis, and treatment of disease, goes back to the 1920s and 1930s, and its growth has been exponential over the last century. This growth is reflected in the value of the global nuclear medicine market, which was worth an estimated $11.77 billion in 2025, growing to a projected $13.72 in 2026 and $66.11 billion in 2034, at a combined annual growth rate (CAGR) of 19.9%.1
According to the World Nuclear Association, each year in the developed world around one person in 50 goes through a diagnostic nuclear medicine process, and approximately one in 500 are treated with radioisotopes.2
This growing market is being driven by greater innovation and by increasing adoption of therapeutic and diagnostic radiopharmaceuticals. Radioligand therapies are a specific subtype of nuclear medicine and combine a radioisotope (radionuclide) with short half-life and a targeting molecule, which are connected by a linker. The targeting molecule, for example a small molecule, peptide, antibody or nanoparticle, directs the radiopharmaceutical to the target tissue, organ, or lesion (see Figure 1). Isotopes with half-lives of three to 10 days allow global distribution to treatment centers from a few or only one manufacturing site. Each dosage must be prepared freshly for the patient and must be administered within the shelf-life.
In diagnosis, healthcare professionals carry out imaging using devices such as gamma cameras, single photon emission computed tomography (SPECT) scans or positron emission tomography (PET) scans. To treat disease, therapeutic radiopharmaceuticals deliver radiation directly to target cells. This localized delivery both reduces systemic exposure and damage to healthy cells, and increases the concentration of isotopes at the target site.3,4,5
Figure 1: Radiopharmaceutical targeting a cancer cell
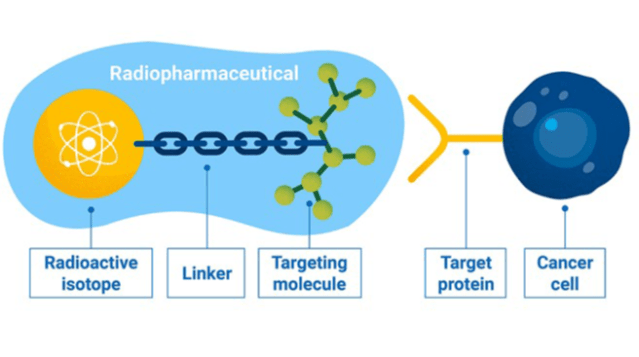
Source: Infographic: A.Vargas/IAEA
Vulnerabilities of the radiopharmaceutical logistic supply chain
Like any other pharmaceutical, radiopharmaceuticals need to be transported safely and in viable condition from the point of manufacturing to the hospital or clinic. They share the logistics challenges of non-radioactive molecules, especially targeted and biological molecules, but bring their own set of challenges as well:
- Time and temperature sensitivity
- Short shelf life of patient individual dosages
- Radiation safety
- Complex global supply chains between isotope manufacturers, radiopharmaceutical manufacturers, and hospitals
- Differences in radiation safety regulations and requirements across countries, airlines, airports and transport hubs.
These challenges make transporting and delivering radiopharmaceuticals a complex process with many potential weak points. Our approach is to focus on precision across the entire logistics chain. This builds in speed, security, care, consistency and dependability, and ensures that the right physician and patient get the right radiopharmaceutical in the right condition and at the right time.
“What makes radiopharma unique is that all of these risks happen simultaneously,” explains Andrea. “You’re managing short half-lives, personalized therapies, pharmaceutical and radiation safety regulatory variations in one integrated workflow. That’s why precision has to be designed into every single step, not added afterwards.”
Putting measures in place for precision in radiopharmaceutical logistics
Radiopharmaceuticals can make huge differences for people with life changing and life limiting diseases, but for these patients, the time to diagnosis and treatment can be critical. Combining this with the short shelf life of a radiopharmaceutical, which may vary from hours to around 10 days4, and the need to manage temperature sensitivity means that we know that speed and efficiency, from manufacturing site to patient bedside, is vital and delays must be mitigated as far as is possible.
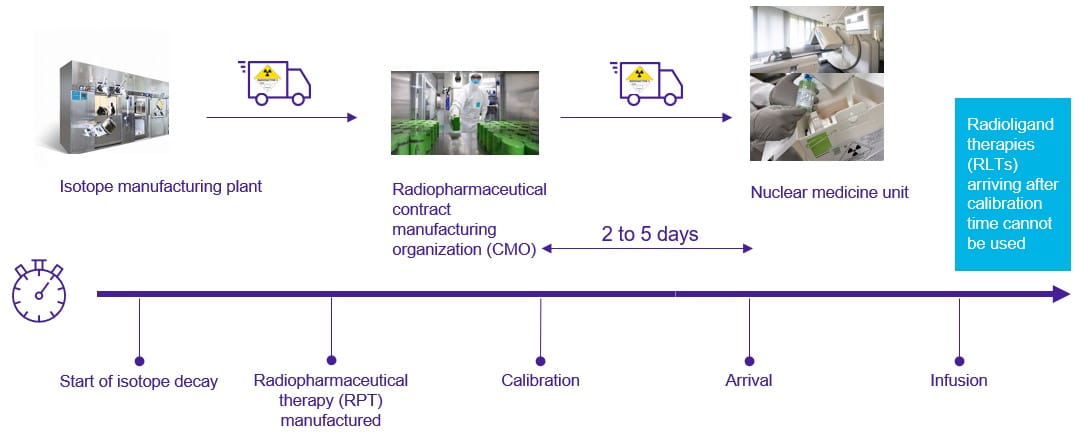
The first step towards this is ensuring collaboration and clear communication between every stakeholder in the supply chain, including manufacturers, shipping teams (land and air), radiopharmacies (also known as nuclear pharmacies), healthcare providers, and clinical/research sites (see Figure 2).
“True coordination means everyone sees the same picture in real time,” says Andrea. “Manufacturers, airlines, ground teams, radiopharmacies and hospitals all need shared visibility. When something shifts — a flight delay, a temperature excursion — we can act immediately rather than react later.”
Targeted diagnostic and therapeutic molecules can also be specific to an individual, so it is essential that they are tracked precisely and get to the right delivery point. We put in place tracking systems so that we can monitor the location, temperature, condition, and radioactivity of the radiopharmaceuticals.
“All of these shipments are prepared for a patient and can only be used in a certain time window,” adds Andrea. “So tracking goes beyond logistics performance — it also encompasses ensuring the therapy reaches the patient at exactly the right moment. That responsibility drives how we design our monitoring and intervention processes.”
This means that we can take measures in real time to ensure that the materials remain viable during their journey and that stakeholders stay safe.
Figure 2: World Courier’s radiopharma logistics process

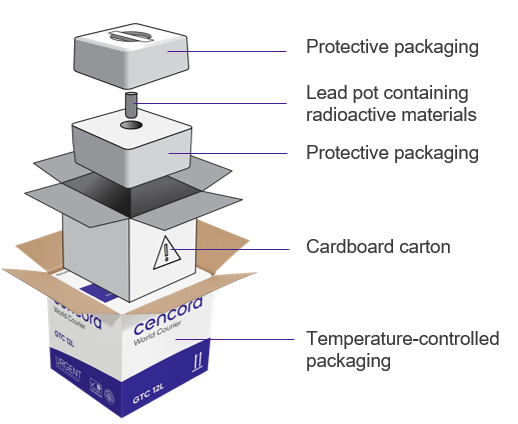
“Radiopharmaceutical packaging is doing two jobs at once,” explains Andrea. “It has to protect the pharmaceutical product from temperature deviation and must fulfill the requirements for radioactive transport packages for radiation shielding and mechanical stability. That balance only comes from experience across thousands of real-world shipments.”
Figure 3: Example of packaging for a radiopharmaceutical (for illustrative purposes only)
Digitization across the supply chain — including real-time shipment monitoring and connected tracking systems — ensures stakeholders have continuous visibility of a radiopharmaceutical’s location and condition throughout transit.
The regulation of radiopharmaceuticals crosses pharmaceutical and radiation safety requirements. Internationally, the International Atomic Energy Agency (IAEA) sets out regulations for the safe transport of radioactive material6, which are then implemented through regional and national frameworks. In the United States, this involves the United States Pharmacopeia (USP), Food and Drug Administration (FDA), Nuclear Regulatory Commission, and the Department of Energy. Variations in how these requirements are interpreted and applied across countries, airlines, airports, and transport hubs can add further complexity to cross-border shipments (see Table 1).
Table 1: Regulatory requirements for radiopharmaceuticals
| Radioactive materials | Pharmaceuticals | Radiopharmaceuticals | |
|---|---|---|---|
| International standards | IAEA | ICH GMP and GCP | Both |
| Regional regulations | IAEA based country regulation | Country and regional GMP, GCP, and GDP | Both |
| Authorities | Country agency for radioactive materials | Country agency for medicinal products | Both |
| License requirement | Country dependent | Always commercial or clinical authorization | Both |
| Packaging | Eight package types based on radiation and use | GMP approved primary and secondary package | Pharmaceutical package in type A package |
IAEO-International Atomic Energy Agency; ICH-International Committee of Harmonization; GMP-Good Manufacturing Practice; GCP-Good Clinical Practice; GDP-Good Distribution Laboratory Practice
“Regulatory alignment is often the hidden challenge,” says Andrea. “Every country, airport and carrier can interpret requirements slightly differently. Our role is to anticipate those variations in advance, so shipments don’t stall when time really matters.”
The role of technology
Artificial intelligence, alongside other advanced digital tools such as digital twins, is increasingly shaping how complex pharmaceutical supply chains are planned and managed. Digital twins create virtual models of logistics networks, allowing teams to explore scenarios, test assumptions, and anticipate potential disruption before a shipment moves. This is an indispensable capability for time critical and highly regulated radiopharmaceutical logistics
“Digital twins allow us to model different scenarios in advance and understand where risk might emerge,” says Andrea. “By stress testing routes, timings, and constraints virtually, we can design more resilient logistics pathways while keeping experienced people firmly in control of every decision.”
Predicting larger-scale disruptive events, such as global pandemics, natural disasters and economic forces, can help to manage risk in the supply chain by simulating risk and modeling its impact.
The future of radiopharmaceuticals logistics
“Ultimately, radiopharmaceutical logistics is about people,” says Andrea. “Behind every shipment is a clinician making decisions and a patient waiting for therapy. Our job is to remove uncertainty from that journey, so science can move faster and care can reach further.”
We continually work to improve the synchronization and efficiency of the entire radiopharmaceutical logistics process. Our proven infrastructure and our collaboration with vetted partners mean that we ensure that the right physician and patient get the right radiopharmaceutical in the right condition and at the right time, every time.
References
- Nuclear medicine market size, share and industry analysis. Fortune Business Insights. 22 December 2025. Available from: https://www.fortunebusinessinsights.com/industry-reports/nuclear-medicine-radiopharmaceuticals-market-101812.
- Radioisotopes in Medicine. Last accessed: 10 January 2025. Available from: https://world-nuclear.org/information-library/non-power-nuclear-applications/radioisotopes-research/radioisotopes-in-medicine.
- Galindo, A., What are Radiopharmaceuticals? IAEA News, 26 February 2025. Available from: https://www.iaea.org/newscenter/news/what-are-radiopharmaceuticals.
- Korde, A., et al., Position paper to facilitate patient access to radiopharmaceuticals: considerations for a suitable pharmaceutical regulatory framework. EJNMMI Radiopharm Chem, 2024. 9(1): p. 2.
- Zhang, S., et al., Radiopharmaceuticals and their applications in medicine. Signal Transduct Target Ther, 2025. 10(1): p. 1.
- Regulations for the Safe Transport of Radioactive Material 2025 Edition, STI/PUB/2120, ISBN 978-92-0-115825-3 Regulations for the Safe Transport of Radioactive Material | IAEA



